FGI requirements for sterile processing facilities
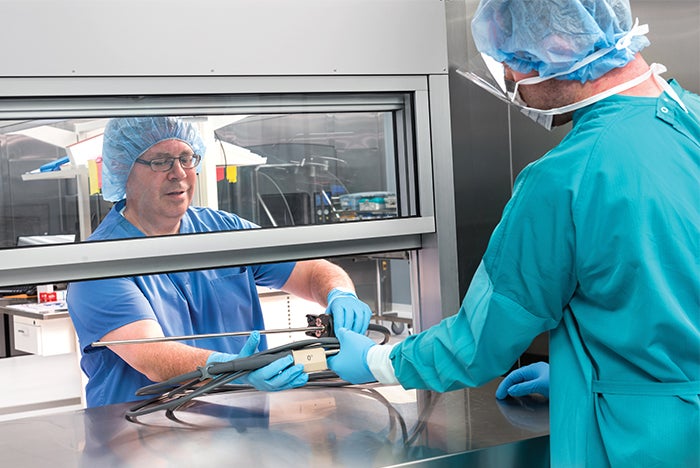
A pass-through window from a decontamination room to a clean workroom.
Image courtesy of Steris Corp.
Sterile processing facilities range from large, highly complex production environments that reprocess hundreds of instrument sets and devices to small facilities dedicated to a specific department or specialty care area that reprocess a limited variety and volume of instruments.
In some situations, sterile processing may take place entirely off-site from where the instruments are used; or small satellite sterile processing facilities may be included in or adjacent to the semi-restricted zone of a surgical suite.
Regardless of the location and variety of reprocessing procedures accomplished in these spaces, the basic operating principles and workflow are the same.
Dirty to clean
A dirty-to-clean workflow is the foundation for design of a sterile processing area. Simply put, it requires a one-way traffic pattern for instruments or devices in which items move from a contaminated state to a decontaminated state.
This one-way workflow is essential to preventing cross-contamination as items move through the process. To support the workflow, ventilation systems are designed to contain contaminants in soiled work areas with negative airflow relative to clean spaces and to protect clean work areas with positive airflow.
While maintaining a dirty-to-clean workflow may seem simple, device-specific cleaning procedures can be complicated and include multiple steps, making these procedures highly error-prone. This is especially true if the sterile processing area does not provide physical separation between dirty and clean areas, appropriate airflow design, and the space, countertops, sinks or other equipment needed to support the correct workflow.
The desire to ensure that facility design supports a dirty-to-clean workflow guided changes to the minimum requirements for sterile processing facilities in the Facility Guidelines Institute’s 2018 FGI Guidelines for Design and Construction of Hospitals and the 2018 FGI Guidelines for Design and Construction of Outpatient Facilities.
The revised requirements, which reflect the FGI Health Guidelines Revision Committee’s (HGRC’s) intent to “support safe environments for patients and staff wherever services are provided,” facilitate the effective reprocessing practices that are critical to prevention of health care-associated infections and thus to patient safety.
Disinfection vs. sterilization
The term “reprocessing” covers a broad set of tasks intended to accomplish one of two levels of freedom from contamination for reusable instruments or devices — either “high-level disinfected” or “sterile.” Whether an item requires high-level disinfection or sterilization is determined by its classification as either a semicritical or critical device per an established guideline known as the Spaulding classification scheme.
Resources
Items that undergo a sterilization process are considered free from all microbial life or “sterile.” However, it is important to understand that items are only considered sterile when all established parameters for validation of the sterilization process have been met and the sterile items are handled and stored in a manner that does not compromise their sterility. The rule for determining whether the sterility of stored sterile goods has been maintained is known as “event-related sterility maintenance.”
The Association for the Advancement of Medical Instrumentation defines event-related sterility maintenance in AAMI ST79, Comprehensive guide to steam sterilization and sterility assurance in health care facilities, as “[s]terility maintenance that is not based on expiration dating but rather on factors such as the quality of the packaging material, the storage conditions, the methods and conditions of transport, and the amount and conditions of handling.” Thus, provision of an appropriately designed sterile storage room is important for maintaining sterility.
In contrast, items that undergo a high-level disinfection process are considered free of all vegetative (living) bacteria, but low numbers of bacterial spores may remain. After undergoing high-level disinfection, such devices must be stored in a manner that keeps them clean and protected from recontamination. Specifically, they must be moved out of or away from the decontamination and high-level disinfection area, dried and stored.
Storage requirements for disinfected items are not the same as for instruments or devices that have been sterilized. Sterilized items must be allowed to cool post-sterilization and then are stored in a clean workroom, clean holding room or sterile storage room that meets HVAC design requirements as described in American National Standards Institute/ASHRAE/American Society for Health Care Engineering Standard 170, Ventilation of Health Care Facilities. Although high-level disinfected items may be stored in these locations, they are not required to be. Instead, these items may be safely stored in a clean cabinet or closed cart.
Whether the final processing step will be high-level disinfection or sterilization, all instrument or device reprocessing begins with the same critical first step: cleaning.
Cleaning is the process of removing bioburden (i.e., viable microorganisms remaining on the surface after use) from a device. Residual bioburden could transfer organisms or pathogens and cause an infection in the next patient on whom the device is used. Inadequately cleaned devices cannot be rendered sterile or effectively high-level disinfected; for this reason, the efficacy of any sterilization or high-level disinfection process depends on consistent procedures, whether manual or automated, for eliminating or significantly reducing bioburden before proceeding to the next processing step.
After cleaning, instruments must be decontaminated. This refers to disinfection by chemical (i.e., disinfectant) or thermal (i.e., washer-decontaminator) means and is intended to render devices safe to handle by staff as they complete the remaining processing steps. Decontaminated instruments are then inspected and packaged for sterilization. Items undergoing high-level disinfection are reprocessed without packaging and thus must undergo a drying process before storage.
Facility requirements
To understand the changes in sterile processing facility requirements in the 2018 FGI Guidelines, it helps to know the history of sterile processing in the surgical suite. The 2010 and earlier editions of the Guidelines required one “substerile” room with a sink and a steam sterilizer between every two operating rooms.
These spaces were intended to facilitate a process known as flash sterilization: a steam sterilization cycle in which unwrapped instruments were subjected to a shortened sterilization cycle for prompt reuse. After the instruments were hand-washed and flash sterilized, they were transported wet in an open pan directly into the operating room. This practice was intended for one or two special instruments or for an instrument accidentally contaminated during a procedure.
As sterilization practices changed, concerns arose about overuse of this process for convenience or as an alternative to purchasing additional instrument sets. In addition, the increasing size and complexity of the instrument sets being flash sterilized and the ability to maintain the sterility of instruments transported wet in an open pan were serious concerns. These issues ultimately led to the elimination of traditional flash sterilization as an accepted practice.
In 2010, the Association for the Advancement of Medical Instrumentation and other professional organizations, including the Association of periOperative Registered Nurses, led a summit to discuss best practices for safely reprocessing instruments needed for immediate use. In a joint statement, the group noted that the term “flash sterilization” was antiquated and would be replaced by a practice that became known as immediate use steam sterilization (IUSS).
IUSS is a rapid turnaround method for sterilization that includes all the aforementioned required cleaning steps and uses closed, protective containers designed and validated for the abbreviated sterilization cycle. Because these rigid sterilization containers protect the sterile item from contamination during transport from the sterilizer to the point of use, it is no longer necessary to build multiple substerile rooms in a surgical suite.
This change resulted in elimination of substerile rooms as a requirement in the 2010 edition of the FGI Guidelines, although the design standards were kept in the document for application when such a room was required by a project’s functional program.
The 2014 edition of the Guidelines introduced the concept of a satellite sterile processing room in or near the surgical suite for facilities that needed rapid turnaround of instruments or IUSS and were not close enough to the main sterile processing department (SPD) to meet these needs. This space was intended to be functionally equivalent to the SPD and required to facilitate a one-way dirty-to-clean traffic pattern.
Where the room was provided, the minimum requirement was for a single room with separate decontamination and clean work areas. This approach established the one-room design as the minimum requirement for a sterile processing room and was based on the belief that either a one- or a two-room sterile processing facility could support a dirty-to-clean traffic flow without risk of cross-contamination.
However, after publication of the 2014 Guidelines, concerns arose that prevention of cross-contamination was difficult in the one-room layout because it wasn’t possible to design an airflow that would ensure that air flowed from the clean side to the dirty side or to ensure that staff could or would maintain the correct workflow in practice.
Two-room solution
To address concerns surrounding the one-room sterile processing facility, the infection preventionists on the 2018 HGRC and other subject matter experts re-evaluated this requirement with an eye to supporting the design of sterile processing facilities that encourage clinical personnel to comply with professional practice guidelines for cleaning, decontaminating and sterilizing surgical instruments.
They considered the importance of the dirty-to-clean workflow in sterile processing areas and determined that the minimum requirement for these spaces should be two rooms: a decontamination room and a clean workroom.
The requirement in the 2018 Guidelines reads, “The two-room sterile processing facility shall consist of a decontamination room and a clean workroom that are physically separated by a wall containing a door or pass-through window that can be closed and secured or a built-in washer/disinfector with a pass-through door or window.”
The 2018 Hospital and Outpatient Guidelines also provide language detailing the minimum physical requirements for both the decontamination room and the clean workroom. Specific requirements such as a three-basin sink with counter enable the required soaking, rinsing and manual cleaning often required for complex instruments along with adequate counter space to set down soiled and clean items separately. Space for sterile storage is also required, either in the clean workroom or in a separate storage room.
The HGRC recognized that in some locations — primarily outpatient areas such as dental clinics — reprocessing is limited to sterilization of single instruments or small instrument sets using a tabletop or similar-sized sterilizer. To allow flexibility for these types of facilities and circumstances, the committee agreed to permit an exception allowing a one-room sterile processing room that supports a dirty-to-clean workflow.
Options for such a design include these two:
- One entrance located approximately equidistant from the clean and decontamination sides of the room to allow for a one-way traffic flow.
- A door at or near the ends of both the decontamination and clean work areas.
Another option for this type of location is to conduct instrument decontamination in a readily accessible, appropriately designed soiled workroom, with packaging and sterilization occurring in a separate clean workroom.
Whether a project includes a one-room or two-room sterile processing facility, the 2018 FGI Guidelines makes it clear that on-site sterile processing facilities “shall be designed to provide a one-way traffic pattern.”
The change introduced in 2014 to move away from provision of substerile rooms between operating rooms was intended to improve practice by reducing the use of flash sterilization. The idea of a one-room sterile processing area to serve the surgical suite at first seemed to answer this need. Time and experience, though, have demonstrated that this concept is not the optimal design to support best practices for sterile processing and was corrected as part of the 2018 Guidelines revision process.
As noted in the “About the Guidelines” introduction to the 2018 edition, “[t]he Guidelines for Design and Construction documents are updated every four years to keep pace with evolving care delivery models, facility types, and requests for up-to-date guidance from care providers, designers, and regulators.”
This change in sterile processing facility requirements is evidence that this process works.
Design evolution
The fundamental steps in reprocessing surgical instruments have remained constant since the early days of steam sterilization.
However, because the complexity and volume of surgical instruments requiring reprocessing have expanded significantly, changes have been required in how these and other instruments are managed in the surgical suite and beyond.
These operational changes have driven the evolution of design requirements for sterile processing facilities wherever they are located.
Paula J. Wright, R.N., CIC, is a project manager in the Infection Control Unit of Massachusetts General Hospital, Boston, and a member of the steering committee of the FGI Health Guidelines Revision Committee. She can be reached at pjwright@partners.org.
ABOUT THIS ARTICLE
This feature is one of a series of articles published by Health Facilities Management in partnership with the Facility Guidelines Institute.





